Learning Hub
Glossary
Machine Bolt
Machine bolts are used to fasten wood to wood, wood to metal, and metal to metal. They are typically installed into pre-drilled and tapped holes and utilized in heavy-duty applications. Earnest Machine stocks Machine Bolts in grades 2, 5, and grade 8, providing options for applications requiring increased strength and durability. Earnest also offers Machine Bolts in various diameters, lengths, and finishes, including RoHS-compliant Zinc Clear Trivalent and hexavalent-free Zinc Yellow Trivalent.

Machine Screw Anchor
A machine screw anchor is a type of fastener used to secure machine screws into masonry or other solid materials like concrete, brick, or stone, where a regular screw wouldn’t hold. It’s essentially a sleeve or plug that gets inserted into a pre-drilled hole in the material. When a machine screw is threaded into the anchor, it expands or grips the surrounding material, creating a strong, reliable mounting point.

Machine Screws
Industrial machine screws are specialized fasteners used to secure components in machinery and equipment. They are designed for precise applications where reliable fastening is required, often involving the assembly of metal parts or the attachment of machine components. These screws come in a variety of sizes, materials, and head styles, making them versatile for different industrial needs.
Machining
A manufacturing process that removes material from a part to shape it into a specific design. In fastener production, machining is used to form precise dimensions, threads, or other features by cutting, drilling, turning, or milling the material with specialized equipment.
MagnaCut Steel
MagnaCut steel, more properly CPM MagnaCut, is a powder-metallurgy stainless tool steel developed specifically for knife blades and other cutting applications where a strong balance of corrosion resistance, toughness, hardness, and edge retention is required. It was engineered to deliver the benefits users often want from premium blade steels without forcing as much of the usual compromise between stain resistance and wear performance. In practical terms, MagnaCut is known for being able to achieve high hardness while still maintaining strong toughness and excellent resistance to rust and staining.
Metallurgically, MagnaCut is designed so that more chromium remains available in solution for corrosion resistance rather than being tied up as large chromium carbides, while vanadium and niobium contribute to wear resistance and edge stability. Because it is produced through powder metallurgy, the carbide structure is finer and more evenly distributed than in many conventional steels, which helps support a combination of strength, sharpenability, and cutting performance. MagnaCut is most commonly associated with premium knives rather than standard industrial fastener or structural applications, so it is generally discussed as a specialty blade steel rather than a general-purpose engineering alloy grade.
Magnesium (Mg)
Magnesium is a silvery-white metallic element with the chemical symbol Mg and atomic number 12. It is a lightweight, strong, and reactive metal that plays a crucial role in both industrial applications and biological systems. As the eighth most abundant element in Earth’s crust and a key component of seawater, magnesium is one of the most important metals for modern manufacturing and life processes alike.
In its pure metallic form, magnesium is one-third lighter than aluminum, making it one of the lightest structural metals. It combines low density, high strength-to-weight ratio, and excellent machinability, which makes it ideal for aerospace, automotive, and electronics industries. Magnesium alloys are often used to produce aircraft parts, transmission housings, laptop casings, and lightweight tools, helping reduce overall weight while maintaining durability.
Chemically, magnesium is highly reactive, especially in finely divided form. It readily reacts with oxygen to form magnesium oxide (MgO), a white powder that emits a bright white flame when the metal burns—this characteristic has made magnesium famous in fireworks, flares, and photographic flashes. When exposed to air, it forms a thin protective oxide layer that prevents rapid corrosion.
Magnesium is also an essential biological element—it is the fourth most abundant mineral in the human body and is necessary for over 300 enzymatic reactions, including energy production, muscle contraction, nerve function, and protein synthesis. It forms the central atom in chlorophyll, the green pigment that allows plants to capture sunlight during photosynthesis.
Common industrial magnesium compounds include magnesium oxide (MgO) used in refractories and insulation, magnesium hydroxide (Mg(OH)₂) used as a flame retardant and antacid, and magnesium sulfate (Epsom salt) used in medicine and agriculture.
Magnesium is typically extracted from magnesite (MgCO₃), dolomite (CaMg(CO₃)₂), and sea water, where it is separated through electrolysis or thermal reduction processes.

Magnesium Oxide (MgO)
Magnesium oxide (MgO) is a white, crystalline, inorganic compound formed by combining magnesium (Mg) and oxygen (O₂). It is a highly stable, non-flammable, and refractory material, meaning it can withstand extremely high temperatures without breaking down. Magnesium oxide occurs naturally as the mineral periclase, but it is most commonly produced synthetically by heating magnesium carbonate (MgCO₃) or magnesium hydroxide (Mg(OH)₂) until they decompose, releasing carbon dioxide or water.

When heated, magnesium reacts with oxygen to form magnesium oxide: 2 Mg + O₂ → 2 MgO
This exothermic reaction produces an intense white flame — a signature of magnesium combustion. The resulting MgO is a fine white powder or solid with a high melting point (~2,852°C or 5,166°F), making it useful in industrial furnaces, crucibles, kilns, and refractory bricks that line high-temperature equipment.
Magnesium oxide has a wide range of uses:
- In metallurgy, it serves as a refractory material and flux, protecting metals from oxidation.
- In construction, MgO boards are used as fire-resistant and mold-resistant alternatives to drywall or cement board.
- In environmental applications, it’s used to neutralize acidic wastewater and flue gases.
- In electrical and thermal insulation, MgO is used inside heating elements (such as in toasters and ovens) due to its high dielectric strength and ability to conduct heat while insulating electrically.
Magnesium oxide also reacts slowly with water to form magnesium hydroxide (Mg(OH)₂), a process known as hydration: MgO + H₂O → Mg(OH)₂
Magnetic Particle Testing (MT)
Magnetic Particle Testing (MT) is a non-destructive testing (NDT) technique designed to detect surface and near-surface defects in ferromagnetic materials like iron, nickel, cobalt, and their alloys. The process begins by magnetizing the part either through direct current or by placing it in a magnetic field. When the material contains a discontinuity—such as a crack, seam, lap, or void—it interrupts the flow of the magnetic field. This disruption causes magnetic flux leakage at the defect site, which provides the basis for detection. Finely milled ferromagnetic particles, applied as either a dry powder or a suspension in liquid, are then spread across the surface. These particles are drawn toward the leakage fields and cluster along the defect, forming a visible indication of its presence.

This method is widely applied across industries like aerospace, automotive, construction, and energy, especially for inspecting welds, castings, and forgings. MT offers several advantages: it is highly effective for finding surface-breaking flaws and shallow subsurface defects, produces immediate and easily visible results, and is relatively quick and inexpensive to perform. However, it does have limitations—it only works on ferromagnetic materials, requires clean surfaces for accurate detection, and may miss deeper flaws that methods like ultrasonic or radiographic testing could identify. Despite these constraints, MT remains a practical, versatile, and widely used inspection tool for ensuring the integrity of critical components.
Magnetite
Magnetite is a naturally occurring iron oxide mineral with the chemical formula Fe₃O₄, and it is one of the most important and magnetic iron ores in the world. It contains both ferrous (Fe²⁺) and ferric (Fe³⁺) iron ions, making it a mixed-valence oxide and giving it unique magnetic properties. In fact, magnetite is the most magnetic naturally occurring mineral on Earth, a property that distinguishes it from other iron ores such as hematite.

In appearance, magnetite is usually black or dark gray with a metallic luster and a high density. When streaked on a surface, it leaves a black streak, unlike hematite, which leaves a reddish-brown one. Its natural magnetism can be strong enough to attract small iron objects or align itself with Earth’s magnetic field, which is why it was historically known as lodestone and used in some of the earliest magnetic compasses.
Geologically, magnetite forms in a wide range of environments. It can crystallize directly from igneous magma, occur as a product of metamorphism, or form through chemical precipitation in sedimentary rocks. It’s commonly found in banded iron formations (BIFs)—ancient layered sedimentary deposits that supplied much of the world’s iron ore. Large magnetite deposits are found in Australia, Brazil, Sweden, Russia, the United States, and South Africa, often mined alongside hematite.
From an industrial standpoint, magnetite is a major source of iron for steelmaking. The ore is typically concentrated using magnetic separation, since its magnetic properties allow it to be easily distinguished and isolated from surrounding rock. After concentration, it is smelted in a blast furnace, where carbon reduces the Fe₃O₄ to metallic iron. The reaction is: Fe₃O₄ + 4CO → 3Fe + 4CO₂
Magnetite is also used in other applications beyond steel production. Finely ground magnetite serves as a dense medium in coal washing, where it helps separate coal from impurities by density. It’s used in catalysts, recording tapes, pigments, water purification, and even in medical applications such as magnetic resonance imaging (MRI) contrast agents and targeted drug delivery systems.
Magni®
A family of engineered coating systems developed by Magni Industries, designed to protect fasteners and metal components against corrosion. Magni® coatings often combine an inorganic zinc-flake basecoat with an organic topcoat, offering customizable friction properties, color options, and high salt-spray resistance for automotive, construction, and heavy-equipment applications.
Major Diameter
The major diameter of a thread is the largest diameter of a screw thread, measured from the crest of the threads on one side to the crest on the opposite side. If you picture the threaded portion of a bolt or screw as a cylinder with ridges cut into it, the major diameter is the diameter of the imaginary cylinder that touches the very tops of those ridges.
For an external thread (such as a bolt), the major diameter is the diameter across the outermost points of the external threads. For an internal thread (such as a nut), it is the diameter measured across the innermost points of the internal thread crests. Because it represents the overall size of the thread, the major diameter is typically the dimension used when identifying or naming a threaded fastener. For example, in an M10 × 1.5 metric bolt, “10 mm” refers to the major diameter.

The major diameter is important because it sets the nominal size of the fastener and influences whether two threaded parts will fit together properly. While the major diameter itself doesn’t carry most of the load—that happens along the flanks at the pitch diameter—it ensures clearance, proper engagement, and compatibility between mating threads.
Together with the minor diameter (measured at the roots of the threads) and the pitch diameter (the effective diameter where threads engage), the major diameter is one of the three critical dimensions that define the geometry and function of a thread.
Manganese (Mn)
Manganese is a hard, brittle, silvery-gray metal with the chemical symbol Mn and atomic number 25. It is a transition metal that plays a crucial role in metallurgy, manufacturing, and biology. While not magnetic in its pure form, it is essential in steelmaking and alloy production due to its strength-enhancing and deoxidizing properties.
In industry, manganese is most widely used as an alloying element in steel—in fact, around 90% of all manganese produced goes into steel manufacturing. When added to iron, manganese improves tensile strength, hardness, and resistance to wear and impact, while also removing oxygen and sulfur impurities during smelting. Manganese steel (also known as Hadfield steel) is famous for its exceptional toughness and wear resistance, making it ideal for railway tracks, rock crushers, armor plating, and heavy machinery.
Manganese is also used in aluminum alloys to increase corrosion resistance and strength, and in batteries, especially alkaline and lithium-ion types, where it appears as manganese dioxide (MnO₂) in the cathode.
Chemically, manganese forms a wide range of oxides and compounds, with oxidation states from +2 to +7, giving rise to colorful salts used in glassmaking, pigments, and fertilizers. For example, potassium permanganate (KMnO₄) is a deep purple compound used as a powerful oxidizing agent in water treatment and chemical laboratories.
Biologically, manganese is an essential trace element required for enzyme function in both humans and plants. It supports bone formation, metabolism, and antioxidant activity, though excessive exposure to manganese dust or fumes (as in mining or welding) can cause neurological disorders known as manganism, similar to Parkinson’s disease.
Manganese occurs naturally in minerals such as pyrolusite (MnO₂) and rhodochrosite (MnCO₃), and major producers include South Africa, Australia, China, and Gabon.
AKA: Hadfield Steel

Marine Hardware
Marine hardware refers to fasteners, fittings, components, and structural accessories designed for use on boats, docks, ships, marinas, offshore equipment, and other environments exposed to water, salt, humidity, UV, vibration, and constant weathering. In simple terms, it is hardware built to survive around water, especially saltwater, where ordinary steel hardware would corrode quickly.
Marine hardware includes a wide range of products such as bolts, screws, nuts, washers, shackles, cleats, eye bolts, turnbuckles, hinges, latches, hooks, rings, rail fittings, chain, wire-rope fittings, dock hardware, brackets, clamps, and mounting hardware. The common thread is that these parts must resist corrosion while also handling mechanical loads, movement, shock, and long-term outdoor exposure.

Material selection is one of the most important parts of marine hardware. Stainless steel, especially 316 stainless steel, is commonly used because it contains molybdenum, which improves resistance to chloride attack from saltwater. 304 stainless steel may be acceptable in freshwater or mild outdoor environments, but 316 is generally preferred for coastal, marine, and salt-spray conditions. Other marine hardware materials may include silicon bronze, phosphor bronze, naval brass, aluminum, galvanized steel, and specialized coated steels, depending on the application.
Corrosion resistance in marine service is not only about “not rusting.” Marine hardware must resist several types of attack, including pitting corrosion, crevice corrosion, galvanic corrosion, and general atmospheric corrosion. For example, stainless steel can perform well when exposed to oxygen, but it may suffer crevice corrosion in tight, wet, oxygen-starved areas. Galvanic corrosion can also occur when dissimilar metals are connected in the presence of an electrolyte, such as saltwater.
In fastener terms, marine hardware often needs careful matching of materials. A stainless bolt installed through aluminum, bronze, or galvanized steel can create corrosion issues if the joint stays wet and electrically conductive. Designers may use isolation washers, sealants, coatings, bedding compounds, compatible alloys, or sacrificial anodes to reduce galvanic corrosion risk.
Marine hardware also has to handle movement. Boats, docks, and marine structures are constantly exposed to vibration, wave action, wind loading, impact, thermal cycling, and repeated wet-dry cycles. That means hardware often needs good fatigue resistance, secure locking features, proper preload, and reliable load capacity. A cleat, shackle, turnbuckle, or eye bolt is not just “holding something”; it may be carrying dynamic loads that change constantly.
Martensite
Martensite is a hard, brittle microstructural phase formed in steel when austenite (the high-temperature FCC phase of iron) is rapidly cooled—or quenched—so quickly that carbon atoms don’t have time to diffuse. Because the transformation happens under diffusionless, high-speed conditions, the crystal structure shifts from face-centered cubic (FCC) to a highly strained body-centered tetragonal (BCT) form rather than transforming into ferrite + cementite (as it would during slow cooling).
This trapped carbon in a distorted lattice creates extremely high internal stress, which gives martensite its defining properties: very high hardness and strength, but low ductility and toughness. That’s why quenched steels are strong but brittle unless further treated.
After martensite forms, steels are typically tempered—heated to a much lower temperature (like 200–600°C)—to relieve stress, reduce brittleness, and allow controlled precipitation of carbides, producing tempered martensite, which offers a better balance of strength and ductility.
Martensite is the foundation of heat-treatable steels used for tools, springs, cutting edges, automotive components, gears, and high-strength fasteners. It is a cornerstone phase in metallurgy because controlling how much martensite forms—and how it is tempered—allows engineers to tune steel performance for extreme mechanical demands.
Martensitic Stainless Steel
Martensitic stainless steel is a category of stainless steel known for its high strength, hardness, and moderate corrosion resistance, achieved through heat treatment that transforms its crystal structure into martensite. It is characterized by a body-centered tetragonal (BCT) crystal structure, which forms when steel containing sufficient carbon is rapidly cooled or quenched from a high temperature. This gives martensitic stainless steels their distinctive ability to be hardened and tempered, much like carbon steels.
The composition of martensitic stainless steels typically includes 11.5% to 18% chromium and 0.1% to 1.2% carbon, with small amounts of other elements such as molybdenum, nickel, or vanadium to improve strength, corrosion resistance, and toughness. The chromium content provides the protective oxide layer that resists rust and oxidation, while the carbon allows for the formation of martensite during quenching, leading to significant hardness and wear resistance.
Unlike austenitic or ferritic stainless steels, martensitic grades are magnetic and can be heat treated to a wide range of hardness levels. However, this comes at a tradeoff: their corrosion resistance is generally lower, especially in environments involving chlorides or acids. They also tend to have reduced ductility and weldability compared to other stainless steel families.
Common martensitic grades include Type 410 (general-purpose, used for cutlery, valves, and turbine blades), Type 420 (high-carbon, used for surgical instruments and knives), and Type 440C (very high carbon, used for bearings, precision tools, and high-wear components). These steels are often used where mechanical strength, edge retention, and wear resistance are more critical than corrosion resistance.
Material Hardness
A material property describing resistance to surface deformation such as scratching, denting, or indentation. It is the underlying property that standardized tests quantify, while the resulting measurement or acceptable range is the hardness level.
Megapascal (MPa)
A megapascal (MPa) is a pressure (or stress) unit in the SI system equal to one million pascals: 1 MPa = 10^6 Pa. Since 1 Pa = 1 N/m² (one newton of force applied over one square meter), MPa is a convenient “bigger” unit for engineering-scale pressures and stresses.
In mechanical and materials work, MPa is especially handy because it lines up cleanly with millimeter-based dimensions: 1 MPa = 1 N/mm². That equivalence comes straight from unit geometry: 1 m² = 10^6 mm², so 10^6 N/m² = 1 N/mm².
Where MPa shows up in industry: you’ll see MPa used for material strength and stress (yield strength, tensile strength, compressive strength, gasket stress/contact pressure), as well as system pressures in hydraulics and process equipment (pumps, pressure vessels, test rigs, fittings). It’s a “sweet spot” unit: Pa is too small for most industrial work, kPa is great for lower pressures (HVAC, pneumatics, weather), and MPa fits nicely for hydraulics and structural/material stress numbers.
MPa is built from two historical pieces. The base unit pascal (Pa) is named after Blaise Pascal, and the official special name “pascal” for N/m² was adopted by the General Conference on Weights and Measures in 1971. The “mega-” part is simply the SI metric prefix meaning 10^6 (one million), so “megapascal” is literally “a million pascals.”
Meissner Effect
The Meissner Effect is the defining feature of superconductivity, describing the phenomenon in which a material completely expels magnetic fields from its interior when it transitions into the superconducting state—that is, when it’s cooled below its critical temperature (Tc).

In ordinary conductive materials (metals like copper or aluminum), magnetic field lines can penetrate the material normally, and cooling them simply reduces electrical resistance without affecting the magnetic field. But in a superconductor, something far more dramatic happens. As the material cools below Tc, it not only loses all electrical resistance—it also actively repels magnetic fields from its interior. This occurs because the superconductor forms surface currents that generate magnetic fields exactly opposing the external field, causing the total internal magnetic field to drop to zero.
This phenomenon was discovered in 1933 by Walther Meissner and Robert Ochsenfeld, who found that magnetic flux lines were expelled from a sample of superconducting tin when it was cooled below its critical temperature in an existing magnetic field. Their observation proved that superconductivity is not merely perfect conductivity (which would allow magnetic fields to remain frozen inside) but rather a distinct thermodynamic phase with unique quantum mechanical properties.
The Meissner Effect is often illustrated through magnetic levitation. When a small magnet is placed above a superconducting material cooled below Tc, the magnetic field lines cannot penetrate the superconductor, and the resulting repulsion creates a stable lifting force that makes the magnet float or levitate. This visible effect is a direct consequence of the perfect diamagnetism of superconductors—that is, their ability to completely cancel internal magnetic fields.
Mathematically, the Meissner Effect is described by London’s equations, which show that magnetic fields decay exponentially from the surface of a superconductor over a characteristic distance called the London penetration depth (λ), typically just a few hundred nanometers. Inside the material beyond this depth, the magnetic field is effectively zero.
Mercury (Hg)
Mercury (chemical symbol Hg) is a heavy, silvery-white metallic element that is unique among metals because it remains liquid at room temperature. It has an atomic number of 80, an atomic mass of 200.59, and belongs to Group 12 of the periodic table, along with zinc (Zn) and cadmium (Cd). The name “Hg” comes from its Latin name hydrargyrum, meaning “liquid silver.”
Mercury is dense, lustrous, and highly mobile, with a melting point of −38.83°C (−37.89°F) and a boiling point of 356.7°C (674°F). It is a poor conductor of heat compared to most metals but an excellent conductor of electricity, which historically made it useful in electrical switches, relays, and thermometers. Because of its ability to form amalgams—alloys with many metals such as gold, silver, and tin—mercury was once widely used in mining, dentistry, and manufacturing.
In the past, mercury had many industrial and scientific applications. It was used in thermometers, barometers, fluorescent lamps, and electrical components, and its vapor was key in early mercury-arc rectifiers and vacuum tubes. However, mercury and most of its compounds are now known to be highly toxic, capable of causing severe neurological and organ damage even at low levels of exposure. For this reason, most of its traditional uses have been phased out or tightly controlled.
Mercury naturally occurs in the Earth’s crust, primarily as the mineral cinnabar (mercury sulfide, HgS), which has a deep red color. It is extracted by heating cinnabar ore in air, which causes the sulfur to combine with oxygen and release elemental mercury vapor, later condensed into liquid form. Small amounts of mercury are also released naturally from volcanic activity and weathering of rocks.
Chemically, mercury is relatively unreactive. It does not oxidize readily in air at room temperature but forms compounds like mercuric oxide (HgO) and mercuric chloride (HgCl₂) under heat or chemical reaction. Mercury can exist in two common ionic states: Hg⁺¹ (mercurous) and Hg⁺² (mercuric), leading to a variety of inorganic and organic compounds—many of which are environmentally persistent and biologically harmful, such as methylmercury (CH₃Hg⁺).
Today, mercury use is heavily restricted due to its toxicity and environmental persistence. Modern technology has largely replaced it with safer alternatives in thermometers, electrical switches, and batteries. However, it still finds limited use in specialized applications such as scientific instruments, dental amalgams, and fluorescent lighting—though even these are declining.

Metal Injection Molding (MIM)
Metal injection molding, often abbreviated MIM, is a manufacturing process used to make small, complex metal parts by combining powdered metal with a temporary binder, injecting that mixture into a mold, removing the binder, and then sintering the part into a dense metal component. It is similar in concept to plastic injection molding, but instead of molding melted plastic into a finished part, MIM molds a metal-powder-filled feedstock that later becomes a solid metal part through thermal processing.
The process begins with very fine metal powder, often stainless steel, low-alloy steel, tool steel, titanium, copper alloy, nickel alloy, cobalt alloy, or another engineered metal powder. That powder is mixed with a binder system made from waxes, polymers, or other temporary carrier materials. The result is a moldable material called feedstock. This feedstock can flow through injection molding equipment and fill a precision mold cavity, allowing manufacturers to create detailed shapes with thin walls, small holes, undercuts, ribs, bosses, threads, splines, and other features that would be difficult or expensive to machine from solid bar stock.
After molding, the part is called a green part. At this stage, it has the correct general shape, but it is oversized and still contains binder. The binder is then removed through a process called debinding, which may use solvent, heat, catalytic chemistry, or a combination of methods. Once most of the binder is gone, the fragile part is called a brown part. It still looks like the intended component, but it has not yet reached final strength or density.
The brown part is then sintered in a controlled furnace atmosphere. During sintering, the metal powder particles bond together at a temperature below the metal’s melting point. The part shrinks significantly as the particles fuse and the voids between them close. This shrinkage is expected and carefully engineered into the tooling design. After sintering, MIM parts can reach high density, often near wrought material density, and can provide strong mechanical properties when the material and process are properly controlled.
Metal injection molding is especially useful when a part is too complex for economical machining but needs to be stronger, tougher, more heat resistant, or more wear resistant than plastic. It is commonly used for medical instruments, firearm components, aerospace hardware, electronics parts, automotive components, lock hardware, cutting tools, gears, brackets, surgical devices, watch parts, and miniature mechanical components.
In fastener and industrial hardware terms, MIM is not typically used for ordinary commodity bolts, nuts, and washers because cold heading, forging, stamping, and machining are usually faster and more economical for those shapes. However, MIM can be valuable for small specialty hardware, complex retainers, inserts, latch parts, clips, locking components, miniature threaded pieces, drive components, and other near-net-shape metal parts where geometry is more important than simple high-volume standardization.
The main advantages of MIM are design freedom, repeatability, reduced machining, efficient material use, and the ability to produce high volumes of complex metal parts. The tradeoffs are tooling cost, process complexity, shrinkage control, size limitations, and the need for careful quality control over powder, binder removal, sintering atmosphere, density, dimensions, and mechanical properties.
Metal Powder Industries Federation (MPIF)
Metal Powder Industries Federation (MPIF) is a not-for-profit trade federation created by the powder metallurgy industry to advance the interests of metal powder producers and metal powder–using industries. It describes itself as a federation of six trade associations covering different parts of powder metallurgy, metal powders, and particulate materials, and it positions itself as a long-time advocate and promoter of PM technology and its industrial benefits.
In day-to-day industry terms, MPIF functions as a hub for technical standards, education, and industry coordination. One of its major technical outputs is its Standard Test Methods for Metal Powders and Powder Metallurgy Products, which provides guidance on terminology and recommended test methods and is developed through committees of technical experts; MPIF also maintains a digital library of technical conference papers dating back decades.
MPIF is headquartered in Princeton, New Jersey (105 College Road East) and supports the industry through member services, publications/news, and events (including conferences that also cover areas like metal additive manufacturing alongside traditional PM).

Metallic Bonding
Metallic bonding is the type of chemical bonding that occurs between metal atoms, characterized by a structure in which positively charged metal ions are surrounded by a “sea” of delocalized electrons. Unlike ionic or covalent bonds—where electrons are either transferred or shared between specific atoms—metallic bonding involves electrons that move freely throughout the metal lattice.
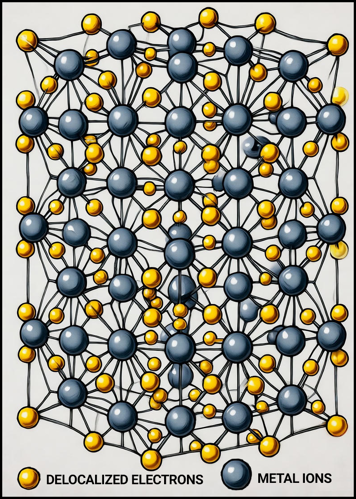
These free electrons act like glue, holding the metal cations together through electrostatic attraction. Because the electrons are not bound to any particular atom, they can move easily through the structure, which gives metals their unique physical properties—such as electrical conductivity, thermal conductivity, malleability, and ductility.
When a force is applied to a metal, the layers of atoms can slide over each other without breaking the bond because the electron “sea” continuously readjusts to maintain attraction between the ions. This is why metals can be shaped and stretched without fracturing.
Metallographer
A metallographer is a materials professional who prepares, examines, and documents the microstructure of metals and alloys so their internal structure, condition, and processing history can be evaluated. In practical terms, a metallographer studies polished and often etched samples under optical or related microscopes to reveal features such as grain structure, phases, inclusions, decarburization, heat-treatment effects, cracks, and other conditions that affect performance. ASM describes metallography as the discipline of examining and determining the constitution and underlying structure of metals and alloys, and it notes the metallographer’s role in helping predict and understand material properties.
The work of a metallographer is both laboratory-based and highly hands-on. It usually involves sectioning a sample, mounting it, grinding and polishing the surface, etching it when needed to reveal structure, then observing, photographing, measuring, and recording the results. In many labs, the metallographer is expected not just to prepare specimens, but also to evaluate what is seen and communicate the findings clearly in reports, especially in quality control, failure analysis, process verification, heat-treat evaluation, and research work.
A metallographer is related to, but not necessarily the same as, a metallurgist. A metallurgist is usually the broader engineering or scientific specialist in metals and alloys, while a metallographer is the person specifically skilled in revealing and interpreting microstructure through specimen preparation and microscopic examination. Some metallographers have formal degrees, while others come up through technical training and extensive practical experience; what defines the role is competence in preparing, examining, documenting, and often interpreting microstructures with limited supervision.
Metalloid
A metalloid is a type of element that exhibits properties intermediate between those of metals and nonmetals. Metalloids have a unique combination of characteristics—some metallic, such as luster, electrical conductivity, and the ability to form alloys, and some nonmetallic, such as brittleness and poor heat conduction. This hybrid nature makes them extremely important in semiconductor technology, alloys, and chemical applications.

On the periodic table, metalloids typically appear along the diagonal “stair-step” line that separates metals (on the left) from nonmetals (on the right). The most commonly recognized metalloids are boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te). Some classifications also include polonium (Po) and astatine (At) depending on their behavior in chemical reactions.
Metalloids often have metallic appearance and moderate electrical conductivity, which can be adjusted by temperature, impurities, or doping—a property that makes them ideal for semiconductors. For example, silicon and germanium are the backbone of the electronics industry, used in computer chips, transistors, and solar cells. Arsenic and antimony, meanwhile, are used to modify alloys, harden lead, or enhance flame retardancy.
Chemically, metalloids can form compounds with both metals and nonmetals, and their oxides often show amphoteric behavior—meaning they can act as either acids or bases. This dual nature reflects their position between the two extremes on the periodic table.
Metallurgy
Metallurgy is the branch of science and engineering that studies the properties, behavior, and processing of metals and their alloys. It involves understanding how the structure of metals—from the atomic level to the macroscopic scale—affects their performance, strength, and durability under different conditions.
The field of metallurgy can be divided into three main areas. Physical metallurgy focuses on how the internal structure of metals (such as grain size, crystal structure, and dislocations) influences their mechanical, electrical, and magnetic properties. Extractive metallurgy deals with the processes used to obtain metals from their ores, including smelting, refining, and electrolysis. Mechanical or industrial metallurgy applies this knowledge to the design, manufacturing, and treatment of metal products—covering processes like casting, forging, heat treatment, and welding.
Metallurgy plays a crucial role in nearly every industry that uses metals, including automotive, aerospace, construction, energy, and manufacturing. By understanding and controlling metallurgical processes, engineers can enhance metal performance, prevent failures such as corrosion or fatigue, and develop new materials with tailored properties for specific applications.
Metric
A system of measurement based on meters, centimeters, and grams, used internationally. In fasteners, “metric” refers to sizes and dimensions measured in millimeters.
Metric Flat Washer
A metric flat washer is a thin, round, flat piece of metal or plastic with a central hole that corresponds to a metric-sized fastener, such as a bolt or screw. It is used to distribute the load of the fastener more evenly across the surface of the material being fastened, preventing damage, reducing friction, and ensuring a more secure connection.

Metric Hardened Flat Washer
A metric hardened flat washer is a type of flat washer designed according to metric dimensions (millimeters) and made from hardened steel. The hardening process involves heat treatment to increase the washer's strength and durability, making it suitable for high-stress applications. These washers are used to distribute the load of a fastener, such as a bolt or screw, over a larger surface area, protect the material being fastened, and reduce wear.

Metric Heavy Hex Nut
A metric heavy hex nut is a type of fastener designed with a hexagonal (six-sided) shape, similar to standard hex nuts, but with a thicker and larger profile. These nuts are commonly used in applications where additional strength and durability are required, particularly in high-load or high-stress environments.
Metric Hex Head Cap Screw
A metric hex head cap screw is a type of fastener that has a hexagonal (six-sided) head and a threaded shaft, designed to be used with metric-sized nuts or tapped (threaded) holes. The hex head allows for easy tightening and loosening using a wrench or socket. These screws are commonly used in machinery, automotive, construction, and structural applications, where precise, strong, and reliable fastening is required.

Metric Hex Head Cap Screw (ANSI)
A Metric Hex Head Cap Screw (ANSI) is a type of threaded fastener with a hexagonal (six-sided) head, used for securing materials or components. It is manufactured to metric dimensions and follows specifications set by the American National Standards Institute (ANSI). These screws are widely used in machinery, construction, automotive, and many other applications.

Metric Hex Jam Nut
Hex Jam Nuts are half the thickness of standard Hex Nuts and are commonly used in applications where a nut with a low-profile thickness is needed. This nut can also be used with a standard Hex Nut to create a locking action in your assembly. Earnest Machine stocks the largest selection of Hex Jam Nuts in industry, offering both inch and metric sizes with a variety of strength levels, thread directions, and finishes to choose from.
Metric Hex Wheel Nut
Otherwise known as Lug Nuts, Wheel Nuts are specifically designed to hold tire rims to an axle. These nuts are primarily used in automotive and construction equipment. In order to match the rim, the nuts are engineered to 60 degree, 90 degree, and radius faces.
Metric Nylon Insert Hex Flange Lock Nut
A metric nylon insert hex flange lock nut is a specialized fastener that combines several important features: a hexagonal shape, a built-in flange, and a nylon insert to provide a secure, vibration-resistant fastening solution. This type of nut is commonly used in applications where both high torque and resistance to loosening are important.

Metric O-Rings
A metric nylon insert hex flange lock nut is a specialized fastener that combines several important features: a hexagonal shape, a built-in flange, and a nylon insert to provide a secure, vibration-resistant fastening solution. This type of nut is commonly used in applications where both high torque and resistance to loosening are important.
Microscrew
A microscrew (miniature screw) is a very small machine screw used to fasten tiny components where space and weight are limited—think electronics, wearables, cameras, watches, and medical devices. In practice, people usually mean sizes ≤ M2 in metric (e.g., M1.6, M1.4, M1.2, M1.0, M0.8) or #0-80 and smaller in inch series (down to #0000-160). They’re made in common head styles (pan, flat, button, socket), drives (Phillips/JIS, Torx®, slotted), and materials like stainless steel, titanium, and brass, with finishes such as passivation or black oxide.
Because everything is tiny, installation uses low, precise torque (often measured in cN·m/N·cm), fine thread classes, and magnified/ESD-safe handling. Variants include thread-forming microscrews for plastics and micro set screws for collars/gears. The key idea: a standard screw in every way—just engineered for microscale assemblies with tight tolerances.

Microstructural Inhomogeneities
Microstructural inhomogeneities are small-scale irregularities or non-uniformities within the internal structure of a material, usually visible under a microscope. Instead of having a perfectly even composition and arrangement of grains, phases, or inclusions, the material contains localized variations that make certain regions behave differently under stress, temperature, or chemical exposure.
These inhomogeneities can take many forms. Common examples include non-metallic inclusions (like oxides or sulfides in steel), segregations of alloying elements during solidification, uneven grain sizes, voids, porosity, or differences in phase distribution (such as harder or softer regions in a heat-treated alloy). They may also include dislocations, twins, or other crystal defects.

While some microstructural variation is normal in engineered materials, significant inhomogeneities often act as weak points. They can disrupt the even flow of stress through the material, leading to stress concentrations that make crack initiation more likely. In fatigue, corrosion, and fracture mechanics, microstructural inhomogeneities are often the hidden initiators behind failure.
Controlling these features is a key part of materials engineering. Careful processing, refining, alloying, and heat treatment are all used to minimize unwanted inhomogeneities and produce consistent, predictable material performance. In advanced applications like aerospace, medical implants, or nuclear power, even tiny microstructural irregularities can be critical to long-term reliability.
Mil-Spec (Military Specification)
Mil-Spec, short for Military Specification, is a formal technical specification created or adopted by the U.S. Department of Defense to define the required characteristics, materials, dimensions, performance, testing, inspection, packaging, marking, or quality requirements for products used in military and defense-related applications.
A military specification is essentially a controlled set of requirements that tells manufacturers, suppliers, inspectors, and buyers exactly what a product must be and how it must perform. Depending on the item, a Mil-Spec may cover the raw material, heat treatment, coating, corrosion resistance, dimensional tolerances, mechanical strength, thread requirements, traceability, environmental performance, test methods, documentation, and packaging. The purpose is to make sure critical parts are consistent, interchangeable, reliable, and suitable for demanding service conditions.
In fastener applications, Mil-Spec fasteners may include bolts, screws, nuts, washers, pins, rivets, inserts, clamps, and other hardware manufactured to military-controlled requirements. These parts are often used in aerospace, defense vehicles, naval equipment, weapons systems, communications equipment, ground support systems, and other assemblies where failure can have serious consequences. A Mil-Spec fastener may need to meet specific tensile strength, shear strength, hardness, plating, passivation, locking feature, thread class, lot traceability, and inspection requirements.
Mil-Spec does not automatically mean “stronger” or “better” in every situation. It means the item conforms to a specific military-defined requirement. One Mil-Spec may describe a high-strength aerospace bolt, while another may describe packaging, labeling, paint, wire, connectors, or test procedures. The value comes from controlled compliance, repeatability, and documented performance, not from the phrase itself.
Military specifications are often identified by prefixes such as MIL-, MS, NAS, AN, or related defense/aerospace standards. For example, an MS part number may refer to a military standard part with defined dimensions and performance requirements, while a MIL-DTL document may describe detailed product requirements. Over time, many older military standards have been revised, canceled, superseded, or replaced by commercial, SAE, ASTM, NAS, or ISO standards, so the exact document revision matters.
In practical industrial terms, Mil-Spec means a product is made, tested, and documented according to a specific military requirement, rather than simply being a generic commercial item. For fasteners, it usually signals tighter control over material, processing, performance, identification, and traceability, especially for defense, aerospace, and other high-reliability applications.
Milling Cutter
A milling cutter is a rotating, multi-edge cutting tool used on milling machines and CNCs to remove material by shearing chips. Unlike a single-point lathe tool or a drill’s two lips, a milling cutter has many teeth that intermittently engage the work, so metal removal comes from the combined action of multiple edges as the tool feeds along a path.
Key anatomy features include the shank or arbor hole, which determines how the tool mounts—either a solid-shank cutter or a shell/face mill mounted on an arbor. The body carries flutes and teeth; the flutes form chip channels, while tooth geometry establishes rake, relief, and helix angle to control chip flow and cutting forces. Cutting diameter and length of cut set the practical step-over and depth capability. Edge style can be square, corner-radius, or ball nose; center-cutting end mills can plunge, whereas non-center-cutting designs cannot.

Common cutter types span end mills (square, ball, corner-radius) for profiling and pocketing, typically with 2–3 flutes for aluminum and 4–6 for steels; face mills or shell mills for planar surfacing using indexable inserts; and slab/plain, side-and-face, T-slot, Woodruff/keyseat, dovetail, and other form cutters that produce specific profiles. Additional families include chamfer and deburr mills, counterbores and countersinks, and thread mills that generate internal or external threads with CNC helical paths.
Cutter materials and coatings are chosen for the job. HSS and cobalt HSS are tough and forgiving but run at lower speeds. Solid carbide is stiff and wear-resistant for high-speed CNC work. Indexable platforms use carbide, ceramic, CBN, or PCD for larger diameters and hard or abrasive materials. Coatings—applied via PVD or CVD—such as TiN, TiCN, TiAlN/AlTiN, and DLC reduce wear and built-up edge; selection should match the work material and expected heat.
Choosing a cutter follows a straightforward logic. By material: aluminum favors high-helix 2–3-flute tools; steels run well with 4–6-flute AlTiN-coated carbide; stainless benefits from a tough grade, sharp edge, and lower helix; titanium and nickel alloys call for variable-pitch carbide and high rigidity. By operation and toolpath: facing suggests a face mill; deep pocketing and 3D work point to a ball or variable-helix end mill; keyways use keyseat cutters; threads benefit from thread milling for better control than tapping. Machine and toolholding matter—stiffer holders (heat-shrink, ER, whistle-notch) and minimal overhang reduce chatter. Chip control depends on flute count and helix for evacuation, and on whether to use coolant, MQL, or run dry, based on the material.
Practical use tips round it out. Prefer climb milling on CNC for better finish and tool life, reserving conventional milling for specific setups. Set feed per tooth (fz) and surface speed (Vc) from manufacturer data, and tune radial (ae) and axial (ap) engagement to avoid chatter. Monitor wear modes: built-up edge suggests raising speed or adding a suitable coating; chipping points to reducing runout or lightening radial engagement; and thermal cracking calls for avoiding intermittent coolant and reducing coolant cycling.
Minimum Breaking Load (MBL)
Minimum Breaking Load (MBL) is the lowest load at which a product is expected to fail (break) when tested to the applicable standard, based on the manufacturer’s rating. It represents a strength threshold, not a recommended working load, and it is typically used as the basis for establishing the Working Load Limit (WLL) by applying a required design factor (for example, WLL = MBL ÷ design factor, when that approach is specified).
MBL is commonly stated for wire rope, chain, slings, synthetic rope, and rigging hardware, and it assumes proper configuration and test conditions. The actual breaking load in a specific test may be higher than the minimum value, but ratings and safety calculations are built around the minimum to ensure consistency. Because configuration and loading conditions matter, MBL can be reduced effectively by factors such as knots, sharp bends (D/d), termination efficiency, wear, corrosion, temperature, shock loading, side loading, or improper thread engagement—which is why WLL and application rules must be followed even if the published MBL looks high.
AKA: Minimum Breaking Strength (MBS), Breaking Load Limit (BLL)
Minor Diameter
The minor diameter of a thread is the smallest diameter of a screw thread, measured from the root of the threads on one side to the root on the opposite side. If you imagine the thread profile as peaks (crests) and valleys (roots), the minor diameter corresponds to the imaginary cylinder that just touches the bottoms of all the thread grooves.
For an external thread (like on a bolt), the minor diameter is the diameter at the base of the external threads. For an internal thread (like in a nut), it is the diameter measured across the crests of the internal threads. In both cases, the minor diameter represents the narrowest point of the threaded section.

The minor diameter is important because it directly affects the strength of the fastener. Since this dimension determines the thickness of the metal left at the root of the thread, it defines how much shear area is available to resist stripping. A larger minor diameter means more material in the cross-section and generally higher strength, while a smaller one means the fastener may be weaker.
Along with the major diameter (measured across the crests of the threads) and the pitch diameter (the effective diameter where the threads of a nut and bolt actually contact each other), the minor diameter is one of the three key dimensions that define a thread’s geometry and ensure proper fit and load distribution.
Mohs Hardness Scale
The Mohs Hardness Scale is a comparative scale that measures the relative hardness of minerals and other materials based on their ability to resist scratching. It was developed in 1812 by German mineralogist Friedrich Mohs and is still widely used today as a quick, practical way to rank material hardness. The scale ranges from 1 to 10, with 1 being the softest and 10 the hardest. Each mineral on the scale can scratch any material ranked lower than itself, but not those higher.
At the soft end, talc is ranked at 1, meaning it can be easily scratched by any other mineral. At the hardest end, diamond is ranked at 10, representing the maximum hardness in natural materials. Other familiar examples include gypsum (2), calcite (3), fluorite (4), apatite (5), feldspar (6), quartz (7), topaz (8), and corundum (9, which includes sapphires and rubies).

The Mohs Scale is not linear but ordinal, meaning the difference in hardness between successive numbers is not equal. For example, diamond (10) is much harder compared to corundum (9) than corundum is compared to topaz (8). Despite this, the scale is practical for fieldwork and industry because it gives a fast and simple way to test hardness using scratch comparisons.
In the context of fasteners and industrial applications, the Mohs Hardness Scale is relevant for understanding wear resistance, abrasion resistance, and material compatibility. For example, coatings, cutting tools, and surface treatments are often selected based on hardness relative to the materials they will interact with. While engineers often use more precise hardness measures such as Rockwell, Vickers, or Brinell hardness tests, the Mohs scale remains a useful reference for comparing material durability in general terms.
Mole
A mole (symbol mol) is the SI unit for amount of substance. It’s basically a counting unit—like “a dozen,” but unimaginably larger.
Formally (SI definition), 1 mol contains exactly 6.02214076 × 10^23 elementary entities. Those “entities” are whatever you specify: atoms, molecules, ions, electrons, or even a defined group of particles.
That exact number is tied to the Avogadro constant N A, which is fixed by definition in the modern SI. So the mole is now defined by an exact count, not by the mass of carbon-12 (older definition).
Molten Aluminium
Molten aluminium is aluminium that has been heated above its melting point until it becomes a liquid metal. Pure aluminium melts at about 660°C / 1220°F, although aluminium alloys may begin melting or soften over a range of temperatures depending on their composition.
In its molten state, aluminium changes from a solid, silvery metal into a bright, fluid liquid that can be poured, cast, alloyed, or processed into new shapes. This is the form used in aluminium casting, where liquid aluminium is poured into molds to make parts such as housings, brackets, engine components, machine parts, structural castings, and other formed products.

Molten aluminium is important because aluminium is lightweight, corrosion resistant, and relatively easy to melt compared with many industrial metals. Steel, for comparison, melts at a much higher temperature, generally above 1370°C / 2500°F depending on the alloy. That lower melting point makes aluminium attractive for foundry work, die casting, recycling, and high-volume manufacturing.
Chemically, molten aluminium is highly reactive with oxygen. When exposed to air, it quickly forms a thin layer of aluminium oxide on the surface. This oxide skin can help protect the metal, but in casting it can also become a defect if it folds into the liquid metal and gets trapped inside the finished part. Foundries manage this through controlled handling, fluxing, degassing, skimming, filtration, and careful pouring practices.
Molten aluminium can also absorb hydrogen, especially when moisture is present. As the metal cools and solidifies, excess hydrogen can form tiny gas pores in the casting. This is why moisture control is extremely important around molten aluminium. Wet tools, damp scrap, or water contamination can be dangerous and can also create serious quality problems.
Molten Zinc
Molten zinc is zinc metal heated above its melting point so it becomes a liquid that can be poured, dipped, or otherwise applied in manufacturing. Zinc melts at about 419.5 °C (787 °F), so “molten zinc” generally means zinc held above that temperature in a controlled pot, kettle, or furnace.

In industrial/fastener contexts, molten zinc shows up most commonly in hot-dip galvanizing, where steel parts are immersed in a bath of molten zinc to form a zinc coating that protects the steel from corrosion. It’s also used in spelter (poured) sockets for wire rope terminations, where molten zinc is poured into a socket to lock the broomed wire strands in place after it solidifies.
Because molten zinc is processed at high temperature, it’s a significant burn and fire hazard, and zinc fumes can be hazardous if inhaled—so industrial practice relies on controlled equipment, ventilation, PPE, and established procedures.
Molybdenum (Mo)
Molybdenum (chemical symbol Mo, atomic number 42) is a strong, silvery-gray metallic element valued for its ability to greatly improve the strength, hardness, and high-temperature performance of steel and other metal alloys. It is often used in small percentages (typically 0.1–1%) but has a major impact on how metals behave under stress, heat, and corrosive environments.
In the context of metallurgy and fasteners, molybdenum plays several crucial roles:
High-Temperature Strength:
Molybdenum has one of the highest melting points of all elements (4,752°F / 2,620°C). When added to steel, it helps the alloy maintain its strength and stiffness even at elevated temperatures. This makes it ideal for automotive, aerospace, and industrial fasteners that must perform under heat and load.
Improved Toughness and Ductility:
It increases the toughness of steel—its ability to deform slightly before breaking—without making it brittle. This means molybdenum-alloyed steels resist cracking or fracturing under shock or cyclic stresses.
Enhanced Corrosion Resistance:
Molybdenum improves resistance to pitting, crevice corrosion, and chemical attack, particularly in chloride or acidic environments. This is why stainless steels containing molybdenum (like 316 stainless) perform well in marine and chemical settings.
Strengthening and Hardening Agent:
It helps form carbides (molybdenum carbides) within steel, which significantly increase wear resistance and surface hardness.
Synergy with Chromium:
When combined with chromium (as in chromoly steel), molybdenum refines the steel’s grain structure, making it stronger, tougher, and more stable under high stress and temperature.
Because of these properties, molybdenum is used in fasteners, turbine blades, furnace parts, engine components, and high-strength tools. In fastener manufacturing, molybdenum-alloyed steels (like 4140 or 8740 chromoly) are common in bolts, studs, and nuts where both strength and reliability under heat are essential.

Monel
Monel is a nickel–copper alloy known for its exceptional resistance to corrosion, high strength, and durability in harsh chemical and marine environments. It typically contains about 65–70% nickel, 20–30% copper, and small amounts of iron, manganese, carbon, and silicon. The combination of nickel and copper produces a material that is stronger than pure nickel, yet retains excellent resistance to acids, alkalis, seawater, and atmospheric corrosion.
Composition and Types
The general formula for Monel alloys includes:
- Nickel (Ni): 63–70%
- Copper (Cu): 20–29%
- Iron (Fe): 1–2.5%
- Manganese (Mn): up to 2%
- Silicon (Si) and Carbon (C): trace amounts
There are several grades of Monel, each tailored for specific uses:
- Monel 400: The most common grade; offers excellent resistance to seawater, hydrofluoric acid, and alkalis, with good mechanical properties from subzero to about 480°C (900°F).
- Monel K-500: A precipitation-hardened version that adds aluminum and titanium for higher strength and hardness while retaining corrosion resistance.
Properties
Monel exhibits several outstanding properties that make it ideal for extreme conditions:
- Corrosion Resistance: Excellent resistance to saltwater, steam, hydrofluoric acid, sulfuric acid, and alkaline environments.
- Mechanical Strength: High strength and toughness, even at low or cryogenic temperatures.
- Nonmagnetic: Monel 400 remains essentially nonmagnetic, which is useful in precision and naval equipment.
- Workability: Can be hot- or cold-worked, machined, and welded, though it’s tougher to machine than standard steels due to its high strength.
Applications
Because of its durability and corrosion resistance, Monel is used extensively in marine, chemical, and aerospace industries:
- Marine engineering: Propeller shafts, seawater valves, pump shafts, and heat exchangers.
- Chemical processing: Equipment for handling hydrofluoric and sulfuric acids, alkali production, and chlorinated solvents.
- Aerospace and defense: Fuel tanks, exhaust systems, and fasteners.
- Oil and gas industry: Cladding for piping, pumps, and downhole equipment exposed to sour gas and brine.
- Musical instruments: Used in trumpet pistons, French horn rotors, and guitar strings for its corrosion resistance and smooth action.
Moon Fasteners
Moon fasteners are half-moon-shaped fastening or retaining components whose name comes from their semicircular profile. The term is not usually a strict, formal fastener classification, but rather a descriptive label applied to hardware that has a rounded half-circle shape and serves a fastening, locating, locking, or retaining function within an assembly. Depending on the application, the term may refer to different types of hardware that share this general shape.
In furniture and wood assemblies, the term may also be used more loosely for half moon nuts, which are also commonly called lunar nuts or crescent nuts. These semicircular threaded components are used with furniture connector bolts to join panels or structural members in items such as beds, tables, cabinets, and other wooden assemblies. They are often installed in a blind bore or recessed hole so the connection is concealed inside the furniture, helping create a cleaner appearance while still providing a strong mechanical joint. In that context, moon fasteners are best understood as semicircular connector hardware used to secure furniture parts together in a compact, partially hidden connection.
In mechanical and power-transmission applications, moon fasteners most commonly refer to Woodruff keys, also known as half-moon keys. These are semicircular keys that fit into a curved keyseat in a shaft and into a matching slot in a mating component such as a gear, pulley, or hub. Their purpose is to help align the parts and transmit torque by locking the rotating component to the shaft. Because of their shape, they seat securely in the shaft and are especially useful where accurate positioning and concentricity matter.
AKA: Half Moon Nuts, Lunar Nuts, Crescent Nuts

Morgan Impact Bend Angle Nail Test
The Morgan Impact Bend Angle Nail Test is a specialized mechanical test used to evaluate the toughness, ductility, and bend resistance of nails. It is designed to determine how well a nail can withstand impact and bending forces without cracking, breaking, or failing prematurely.
In the test, a nail is driven against or bent over a specified angle using a controlled impact load, often with a pendulum or hammer-like device. The nail is struck until it bends to a predetermined angle, and the degree of bending and the condition of the nail afterward (whether it bends smoothly, fractures, or shows signs of brittleness) are recorded.
The purpose of the Morgan test is to replicate the kinds of stresses a nail might face during real-world use — for example, when being hammered into tough materials or subjected to repeated impact in structural applications. A nail that passes the test demonstrates good impact resistance, bending capacity, and toughness, qualities important in construction, woodworking, and heavy-duty fastening.
This test is often cited in quality control and standardization for nail manufacturing, as it helps ensure that nails meet performance requirements for durability and safety.


