Learning Hub
Glossary
Galvanizing Process
The galvanizing process for steel materials begins with caustic cleaning (degreasing), where steel parts are immersed in a hot alkaline solution, typically caustic soda. This step removes oils, grease, dirt, and organic contaminants from the surface. The parts are then rinsed in water to eliminate residues before moving to the next stage. After degreasing, the steel undergoes pickling (acid cleaning), which involves dipping the parts into a dilute acid bath—commonly hydrochloric acid (HCl) or sulfuric acid (H₂SO₄). This treatment removes mill scale, rust, and oxides that remain on the steel surface. Another water rinse follows to prevent acid carryover into later steps.
Once the steel is clean, it enters the fluxing stage. Here, the steel is dipped into a flux solution, typically zinc ammonium chloride (ZnCl₂ + NH₄Cl), which coats the surface. The flux prevents oxidation before immersion in molten zinc and helps ensure proper bonding between the steel and zinc during galvanizing. In some cases, a dry flux layer is applied and dried on the steel surface. When this method is used, a drying step may follow to reduce spattering when the steel is submerged into the molten zinc bath.
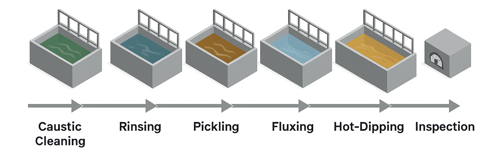
The core of the process is hot-dip galvanizing, where the steel is immersed in a bath of molten zinc maintained at about 450 °C (840 °F). At this temperature, the zinc metallurgically reacts with the steel surface, forming a series of zinc-iron alloy layers that are tightly bonded to the steel, topped with an outer layer of pure zinc. This metallurgical bond makes the coating exceptionally durable and resistant to peeling or flaking. After being withdrawn from the zinc bath, the parts undergo cooling and solidifying, either by air cooling or water quenching, which hardens the coating and prepares the galvanized product for handling.
Finally, the parts undergo inspection to verify the quality of the coating. This includes checking for coating thickness, adhesion, and uniform coverage. Inspections are carried out through visual examination, thickness measurements, and adherence testing to ensure the finished galvanized coating meets industry standards. The end result is a galvanized steel product with a protective zinc coating that provides both a physical barrier and sacrificial protection against corrosion, significantly extending the material’s service life in harsh environments.
